Spin‑spray‑assisted layer‑by‑layer (LbL) assembly is an innovative technique for producing nanostructured thin films due to its rapid deposition and excellent substrate coverage. In this article, Farid Fadhillah fabricated a nanofiltration (NF) membrane composed of multilayers of polyethyleneimine (PEI) and poly(sodium‑4‑styrene sulfonate) (PSS) on a polysulfone (PSF) support. The resulting membrane was subsequently coated with a metal–organic framework (MOF303).
The fabricated (PEI/PSS)₅–MOF303 membrane demonstrated a rejection rate of 18.94 ± 1.58% and a permeability of 0.91 ± 0.13 L/(h·bar·m²), while also exhibiting improved antifouling performance. These findings highlight the potential of spin‑spray‑assisted LbL assembly as a promising route for thin‑film composite membrane fabrication.
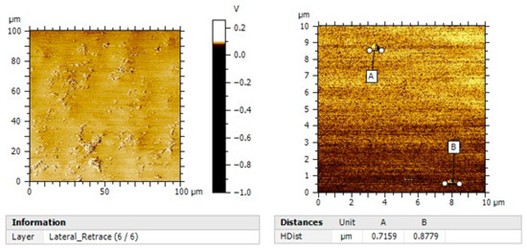
Surface characterization was performed using a commercially available AFM system equipped with a NanoWorld Arrow‑CONTR AFM probe, a silicon cantilever with a force constant of 0.2 N/m, operated in contact mode. Lateral images were used to visualize surface inhomogeneities across the scanned region. The NanoWorld AFM probe ensured stable tip–sample interaction, enabling high‑quality topographical and lateral force mapping. This article emphasizes the importance of selecting a reliable AFM probe for nanoscale membrane characterization.
Full Citation:
Farid Fadhillah. Fabrication of Thin-Film Composite Nanofiltration Membrane Employing Polyelectrolyte and Metal–Organic Framework (MOF) via Spin-Spray-Assisted Layer-by-Layer Assembly. Engineering Proceedings, 2025, 105(1). DOI: https://doi.org/10.3390/engproc2025105003
Citing Licence
This article is published under the Creative Commons Attribution (CC BY) license, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. © 2025 by the author. Published by MDPI.