Gram-negative bacteria release outer membrane vesicles (OMVs) that play a central role in host–pathogen interactions by transporting biomolecules, including proteins and nucleic acids. In this article, Marisela Velez and Véronique Arluison investigate the role of the RNA chaperone Hfq in mediating the interaction of small regulatory RNAs (sRNAs) with bacterial membranes.
In this article, it is shown that RNA binding to the inner membrane of Escherichia coli occurs in an Hfq-dependent manner. The study further demonstrates that membrane composition is a key factor in this process, with cardiolipin-rich lipid domains significantly enhancing RNA–membrane interactions. These findings provide new insight into the mechanism of RNA translocation from the cytoplasm to the periplasm, supporting its subsequent incorporation into OMVs.
Atomic force microscopy (AFM) was used to verify the formation and integrity of supported lipid bilayers and to monitor peptide–membrane interactions. Imaging was performed in tapping mode using a NanoWorld PNP-DB AFM probe with a resonance frequency of 15 kHz and a spring constant of 0.48 N/m. Measurements were carried out in liquid environment, enabling high-resolution characterization of biologically relevant membrane structures.
This work highlights the importance of AFM-based analysis for studying lipid–protein interactions and provides new understanding of RNA transport mechanisms in bacterial systems.
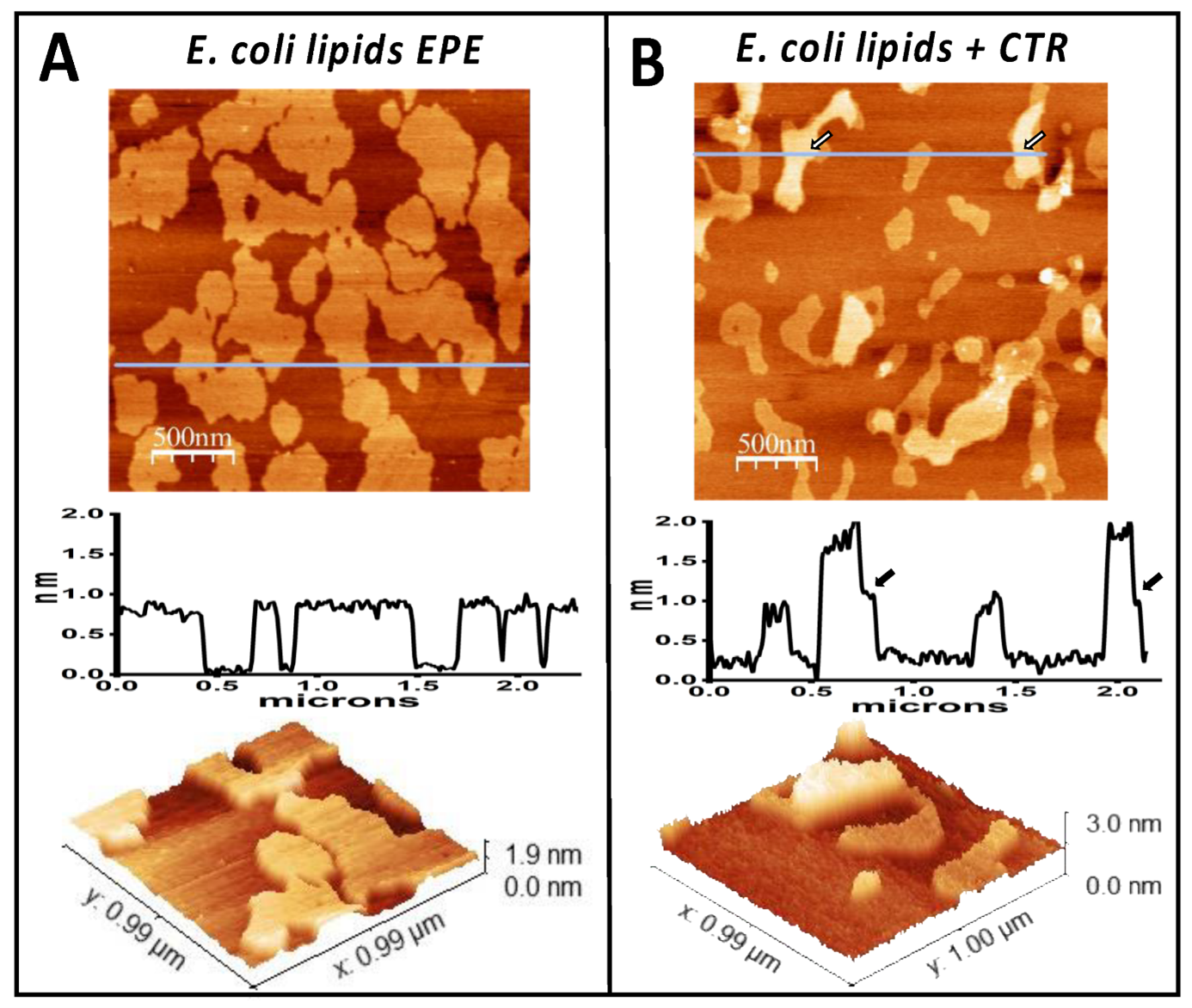
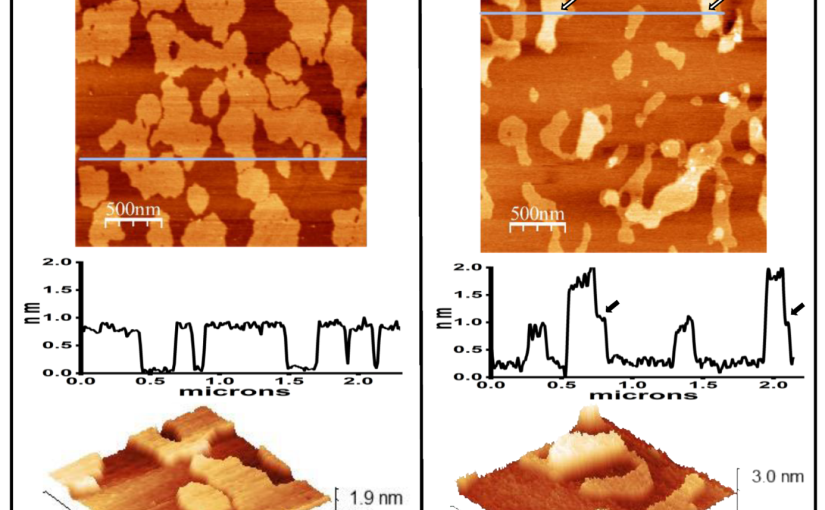
E. coli lipid bilayer incubated in the absence (A) or presence (B) of Hfq-CTR. Panel (A) shows the E. coli lipids bilayer. The height profile under the line shown on the upper image indicates that the domains are 0.8 nm higher than the rest of the membrane. The lower panel shows a three-dimensional representation of a small region. Panel (B) shows the E. coli lipid bilayer incubated in the presence of Hfq-CTR. The peptide accumulated on top of some of the domains, generating 1 nm high regions in some of them, as shown on the height profile. The arrows point the regions where the change in height occurs.
Full citation:
Velez, M.; Arluison, V.
Does the Hfq Protein Contribute to RNA Cargo Translocation into Bacterial Outer Membrane Vesicles?
Pathogens 2025, 14(4), 399.
https://doi.org/10.3390/pathogens14040399
License: CC BY 4.0 (https://creativecommons.org/licenses/by/4.0/)