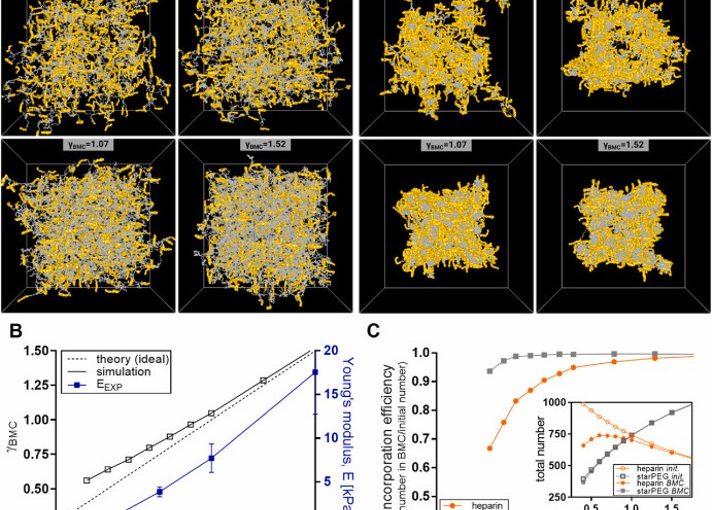
Glycosaminoglycan-based biohybrid hydrogels are highly promising materials for tissue engineering and regenerative medicine due to their ability to provide cell-instructive environments. In this article, Jana Sievers-Liebschner, Ron Dockhorn, Jens Friedrichs, Thomas Kurth, Peter Fratzl, Jens-Uwe Sommer, Carsten Werner, and Uwe Freudenberg investigate the nanoscale molecular network structure of these hydrogels using an integrated analytical approach.
The study combines transmission electron microscopy, X-ray scattering, computer simulations, and AFM-based nanoindentation to quantitatively characterize nanoscale polymer network connectivity and structural inhomogeneities. These parameters are essential for understanding hydrogel mechanics, growth factor delivery, and cell–material interactions relevant to regenerative therapies and organoid culture systems.
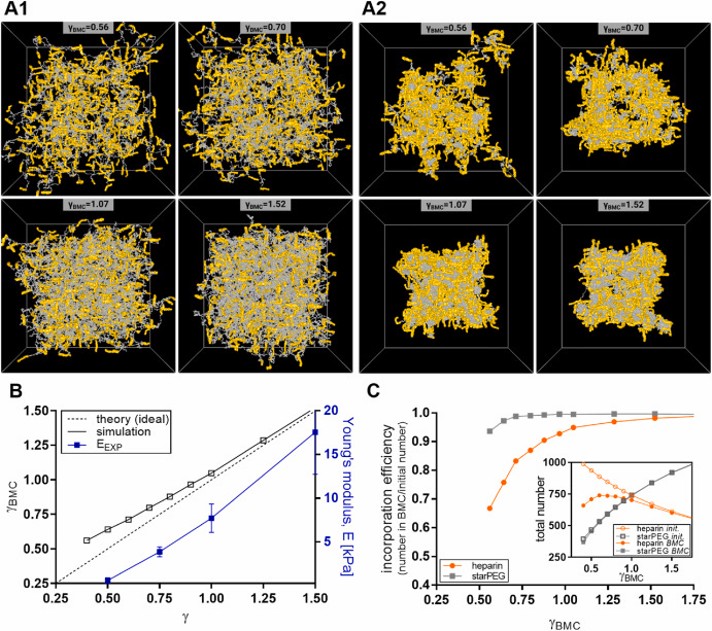
Atomic force microscopy (AFM)-based nanoindentation measurements were performed to determine the mechanical stiffness of the hydrogels in both PBS and ethanol environments. Measurements were conducted using a modified NanoWorld PNP-TR-TL-Au AFM probe equipped with a 10 μm silica bead for colloidal probe nanoindentation.
Nanoindentation experiments were carried out using a set point of 6 nN and an approach/retract velocity of 5 μm/s. At least 70 force–distance curves were recorded for each sample at different positions across the hydrogel surface. Young’s modulus values were extracted using the Hertz model, enabling quantitative evaluation of hydrogel nanomechanical properties.
This work demonstrates how AFM-based nanoindentation with a NanoWorld AFM probe contributes to the detailed characterization of biohybrid hydrogel networks and supports the development of engineered matrices for biomedical applications.
Full citation:
Sievers-Liebschner, J.; Dockhorn, R.; Friedrichs, J.; Kurth, T.; Fratzl, P.; Sommer, J.-U.; Werner, C.; Freudenberg, U.
Unravelling the molecular network structure of biohybrid hydrogels.
Materials Today Bio 2025, 34, 102249.
https://doi.org/10.1016/j.mtbio.2025.102249
Copyright © 2026 Elsevier B.V.